More than 1 million people worldwide acquire a sexually transmitted infection (STI) every day, many of which occur without symptoms1. There is little difference in the prevalence of sexually transmitted infections between rural and urban communities transnationally2. Worldwide, there are inequalities in STI testing; in some rural areas, testing is less frequent than in urban environments despite higher local STI prevalence3.
Access to health services in rural areas is usually more challenging than in urban areas4. In many countries, health care and health outcomes suffer as a result5. Barriers to accessing sexual health services present even greater challenges due to the sensitivity of the topic6-8.
Despite the launch of the Scottish government strategy in 2003 to enhance sexual wellbeing, the rate of STIs continues to increase9. Chlamydia trachomatis is the most prevalent bacterial STI in Scotland, with 17 371 cases of infection diagnosed in 2013, a rise from 12 288 in 2002. However, the last two years have shown a decrease in infection rate10. Chlamydia infection can lead to complications such as infertility, pelvic inflammatory disease, epididymo-orchitis (men) and reactive arthritis9-12. It is for these reasons, as well as the large number of tests, that Chlamydia was used to study accessibility to STI testing.
In rural communities, maintaining confidentiality is challenging13. Consulting a local GP or pharmacist can be difficult because of tight social networks and perceived stigma6-8. Furthermore, people in rural communities have much further to travel for an anonymous STI testing service, although postal kits may have some advantages in this situation8,14.
The pattern of testing in remote and rural areas is poorly understood. It is possible to speculate about the barriers to accessing sexual health and family planning services but there is only a limited picture of what these barriers are8. While there is some research in developing countries, there has been little work in very remote areas in developed countries8,10,15. The Highlands of Scotland present a good opportunity to assess these issues.
The aims of the project were therefore to quantify the incidence of Chlamydia infection diagnosed in remote communities, assess where people in remote and rural communities go to access testing, and the type of test they opt for.
Retrospective data were collected from four sources for Chlamydia tests over a 1-2 year period between 2012 and 2014. All results processed through the Raigmore Hospital Microbiology Laboratory in Inverness, which is the main testing centre for North and West as well as South and Mid Highland, were looked at.
To analyse where a test was taken in relation to where the patient was from, postcode data were required. Postcodes were given a marker of 'rurality' using the Scottish Government Urban/Rural classification 8-fold index (UR8)16 as well as an index of deprivation using the Scottish Index of Multiple Deprivation Quintile (SIMD)17.
Due to the use of full postcodes, which could potentially identify individuals, ethical approval was sought and approved from both the National Research Ethics Service (NRES) committee and the Caldicott approval (UK process for safeguarding the confidentiality of patient data). Once the data had been given SIMD and UR8 markers, the full postcodes were discarded and the results effectively anonymised. For data from Highland Brook (a sexual health service in Inverness for people under 25), Highland Sexual Health and postal testing, manual decoding of postcodes was done by the organisations themselves.
All of the test results used in the study were present in the data set from the microbiology laboratories, whether taken from a GP practice, postal tests or family planning results. Family planning test results appeared in the microbiology data set as anonymised results without postcodes attached. It was only possible to access results with the postcodes attached directly from the organisations themselves. To avoid double counting these results, the family planning and postal testing results from the microbiology data set were removed. Postal test results were identified within the microbiology data set by identifying results with matching postcodes and were done within 7 days pre/post the microbiology tests.
A total of 9644 results were then used for the final analysis. A database was created to show test date, result, patient SIMD and UR8 and 'test' SIMD and UR8 and type of test and analysed using 'python'.
Ethics approval
Ethics approval was granted by NHS Research Ethics Committee (NREC) North of Scotland, NHS Research Ethics Committee (#13/NS/0118). The IRAS project ID number was 128063.
A total of 9644 results were analysed from results from 2012 to 2014 (Table 1).
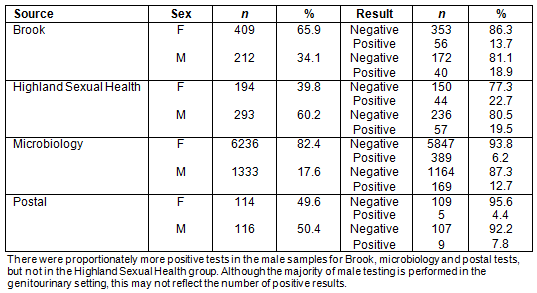
A total of 77% of the results were female tests and 22% were male. For Brook and microbiology, most tests were female tests. Most of the results from the NASH (National Sexual Health System) database from Highland Sexual Health were male. For the postal testing, tests were used by men and women equally.
Overall, 8.1% of the results were positive and 84.4% of the results were negative. A small proportion of all tests was rejected due to a problem with the way the sample was taken (1%) or labelled, and some were rejected due to a problem with the test result (6.5%), described as an indeterminate result by the laboratory. There were proportionately more positive tests from STI clinics (Highland Sexual Health and Brook), compared with microbiology tests and postal tests.
The proportion of men who had positive tests was almost double that for women although men made up only 27.9% of the total number of tests (odds ratio 2.14, p<0.0048) (Table 2). The majority of male testing was performed in a genitourinary setting; however, this was not reflected in the number of positive results (Appendix I).
Table 1: Proportion of male and female tests
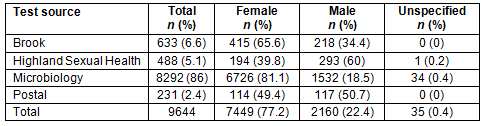
Table 2: Distribution of positive and negative test results

SIMD quintiles
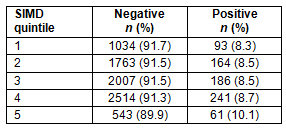
Positivity was assessed in relation to the SIMD quintile17 (Table 3). There were 724 results that were missing either a positive or a negative test result. There were 370 results that were missing the patient SIMD. A total of 8606 results were analysed in relation to SIMD, with 1056 results not used because they were missing either a result or the patient's residence; hence, the SIMD could not be calculated.
Chi-squared analysis showed that there was no significant difference between the SIMD quintile of a patient's home and whether their test result was positive or negative (χ2(4)=1.99; p=0.73), with the likelihood of a positive or negative test similar across all categories.
Table 3: Distribution of positive and negative results according to Scottish Index of Multiple Deprivation Quintile (where quintile 1 is least deprived and quintile 5 most deprived)
Urban Rural 8-fold index
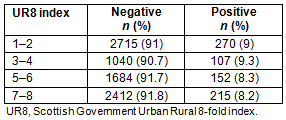
The rate of positivity was assessed in rural compared with urban settings using the Scottish Government UR8 classification16 (Table 4). A total of 8585 results were analysed in relation to UR8, with 1067 results not used because they were missing either a result or the patient's residence; hence, the UR8 could not be calculated.
Chi-squared analysis showed that there was no significant correlation between the rurality of a patient's home and the likelihood that their test result would be positive or negative (χ2(3)=2.30; p=0.510, with the likelihood of a positive or negative similar across all categories.
Table 4: Distribution of positive and negative results according to Urban Rural 8-fold classification (where 8 is the most rural and 1 is the most urban)
Where people are tested
Where people are tested in relation to where they live was assessed: 383 results were missing the patient UR8 and 166 results were missing the test UR8. A total of 536 results in total were missing either one or the other; 13 were missing both. In total, 9126 results were assessed (Table 5). A 'rural' postcode was classified as UR8 7-8; 'urban' was classified as UR8 1-6.
Over one-third (37.7%) of patients living in UR8 7-8 (rural) had their test done in a UR8 1-6 (urban) area.
A similar set of analyses using a dichotomous urban/rural classification, with urban being represented as UR8 1-2, showed that 537 tests of the total 2627 patients in UR8 7-8 (20.4%) had their test performed in a UR8 1-2 area. These results can also be broken down to identify their test location.
A secondary analysis assessed where people from rural areas who ultimately tested positive had their tests performed. They were probably more likely to have gone to an urban test centre than those who tested negative when comparing UR8 7-8 areas with UR8 1-6 (Table 7).
Table 5: Using Urban Rural 8-fold classification to look at where people are tested, comparing the most remote (UR8 7-8) with more urban (UR8 1-6)
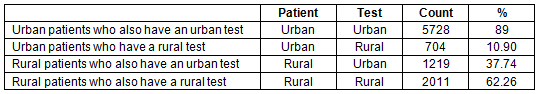
Table 6: Distribution of positive and negative results and test location amongst people living in very rural areas (UR 7-8) who have had their test in a very urban centre (UR 1-2)
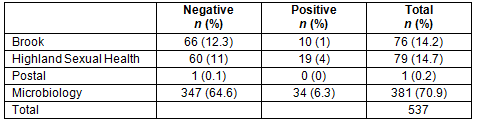
Table 7: Distribution of positive and negative results amongst rural patients who have their test in a rural setting and rural patients who have their test in an urban setting

Discussion
Main findings
Most (77.2%) of the Chlamydia tests were performed on women. This reflects the national Scottish statistics for Chlamydia testing where 73% tests are performed on women18. There were proportionately more male tests in the Highland Sexual Health service as well as almost equal numbers of male and female tests through postal testing. Both of these testing methods are anonymous. This may suggest that men are more likely to opt for anonymous testing. This also reflects National Health Service (NHS) data that show that the majority of male testing is performed in genitourinary medicine settings18 as well as a Scottish study that showed that postal testing was the most frequent way men submitted a test beyond the genitourinary medicine setting19. The proportion of men who had positive tests was almost double the proportion for women, although men made up only 28% of the total number of tests. This is a similar finding to the NHS Information Service Division findings within NHS Highland for 2010 and would suggest that future testing could be better at targeting men18.
There was no significant difference in positivity between rural and urban areas, reflecting previous work2, although these findings are new for Scotland. This has implications in that rural areas may require more attention when it comes to provision of resources because there is as high a proportion of people from rural areas with positive tests as from urban areas. Similarly, there was no significant difference in positivity when comparing with the SIMD. This is different from nationwide studies across England where there was an increase in STI rate in more deprived areas19. It could be speculated that this difference may be because deprivation is more difficult to measure accurately in remote communities20.
Many people travel for their tests: 37% of people from very rural areas (UR8 7-8) had their test done in a more urban (UR8 1-6) setting and 20% were tested in a very urban setting (UR8 1-2). This implies that a large number of people travel for their tests. This could be because their nearest health centre is in an urban area, they had to have the test done in an urban area for another reason (eg gynaecology clinic, patient in hospital) or this may be where they work/go to school etc. It could also be that they went to an urban area because they knew they could have an anonymous test. They may have attended a family planning clinic because they had symptoms, supporting the higher rate of positive tests in travellers. Further assessment is not possible from these data, but could be gathered through further qualitative research. This study shows that a large proportion of people living in the most remote areas travel to more urban areas to have their test and there may be more positive tests among rural people having their test in an urban setting. Overall, this may have implications for increasing provision of sexual health services but particularly for the provision of anonymous testing to remote and rural areas.
This work largely confirms findings in previous studies. The National Scottish Statistics for Chlamydia testing also found that most testing is being performed on women, and also that men mostly opt for testing in a genitourinary setting18. Previous studies have also found that men are more likely to opt for a postal test and that men also have more positive tests than women18,21. More positive tests coming from the genitourinary setting overall also reflects previous studies22,23 as well as no significant difference in positivity between rural and urban areas2.
Strengths
This study has looked at 9644 tests. As far as the authors are aware, this is the largest study of its kind. This study has also been able to look at patients' home postcodes in relation to where they have their test, using a full postcode, which allows a detailed look at the effect of rurality on testing.
Limitations
Although the results might be a true reflection of testing in Scotland, they may not hold true for other rural areas or countries because systems and cultures may differ. To establish testing for all of remote and rural Scotland, other testing laboratories would need to be included because some centres send results to Glasgow. Some people in the regions studied here may also go to other cities for testing.
In contrast to the findings of previous work that shows that there are more positive tests in deprived areas19, this study found no difference in positivity between rural areas and SIMD. Many people travel for their tests in rural Scotland and, at least in this setting, there may be a higher proportion of positive tests from those who travel furthest.
It would be valuable to take this study further in four ways. First, replication of the findings in other countries would be valuable. Second, qualitative interviews with young men and women from rural communities about barriers to accessing sexual health services would help to gain a more in-depth understanding of rural barriers to accessing services. Third, use of Geographical Information Systems software to examine distance from testing locations would give a greater understanding of the geographical factors involved in decisions about place of testing. Finally, this study only looks at STI testing. It would be valuable to examine the role of geography in use of family planning services and sexual health in a more general way.
References
1. WHO. Sexually Transmitted Infections. Factsheet 110 (Internet) 2013. Available: http://www.who.int/mediacentre/factsheets/fs110/en (Accessed 11 April 2015).
2. Winter L, Goldy AS, Baer C. Prevalence and epidemiologic correlates of Chlamydia trachomatis in rural and urban populations. Sexually Transmitted Diseases 1990; 17(1): 30-36.
3. Ferris DG, Litaker M. Chlamydial cervical infections in rural and urban pregnant women. Southern Medical Journal 1993; 86(6): 611-614.
4. WHO. Increasing access to health workers in remote and rural areas through improved retention. Global policy recommendations (Internet) 2010. Available: http://www.who.int/hrh/retention/guidelines/en (Accessed 11 April 2015).
5. Strasser R. Rural health around the world: challenges and solutions. Family Practice 2003; 20(4): 457-463.
6. Richardson D, Maple K, Perry N, Ambler E, Jurd C, Fisher M. A pilot qualitative analysis of the psychosocial factors which drive young people to decline Chlamydia testing in the UK: implications for health promotion and screening. International Journal of STD and AIDS 2010; 21(3): 187-190.
7. McNulty CAM, Freeman E, Bowen J, Shefras J, Fenton KA. Barriers to opportunistic chlamydia testing in primary care. British Journal of General Practice 2004; 54(504): 508-514.
8. Docherty A. Accessing sexual health services in rural Scotland. British Journal of School. Nursing 2010; 5(2): 78-86.
9. Geisler WM, Wang C, Morrison SG, Black CM, Bandea CI, Hook EW 3rd. The natural history of untreated Chlamydia trachomatis infection in the interval between screening and returning for treatment. Sexually Transmitted Diseases 2008; 35(2): 119-123.
10. Health Protection Scotland. Genital Herpes simplex, genital Chlamydia and Gonorrhoea infection in Scotland: laboratory diagnoses 2004-2013 (Internet) 2014. Available: http://www.hps.scot.nhs.uk/bbvsti/wrdetail.aspx?id=60636&wrtype=9 (Accessed 10 April 2015).
11. Bhattacharya S, Porter M, Amalraj E, Templeton A, Hamilton M, Lee AJ et al. The epidemiology of infertility in the North East of Scotland. Human Reproduction 2009; 24(12): 3096-3107.
12. Wallace LA, Scoular A, Hart G, Reid M, Wilson P, Goldberg DJ. What is the excess risk of infertility in women after genital chlamydia infection? A systematic review of the evidence. Sexually Transmitted Infection 2008; 84: 171-175.
13. Townsend T. Ethics conflicts in rural communities: privacy and confidentiality. In: WA Nelson (Ed). Handbook for rural health care ethics: a practical guide for professionals. Lebanon, NH: Dartmouth College Press, 2009.
14. Senok A, Wilson P, Reid M, Scoular A, Craig N, McConnachie A et al. Opportunistic screening for genital chlamydial infection. Journal of Epidemiology and Community Health. 2005; 59: 198-204.
15. Quine S, Bernard D, Booth M, Kang M, Usherwood T, Alperstein G et al. Health and access issues among Australian adolescents: a rural-urban comparison. Rural and Remote Health (Internet) 2003; 3(3): 245. Available: www.rrh.org.au (Accessed 10 July 2015).
16. Scottish Government. Urban Rural Classification 2011-2012. (Internet) Available: http://www.scotland.gov.uk/Topics/Statistics/About/Methodology/UrbanRuralClassification/Urban-Rural-Classification-2011-12/2011-2012-Urban-Rural-Lookups. Accessed 28 March 2015.
17. Information Services Division Scotland. Scottish Index of Multiple Deprivataion. ISD Scotland (Internet) 2010. Available: http://www.isdscotland.org/Products-and-Services/GPD-Support/Deprivation/SIMD (Accessed 29 September 2014).
18. Information Services Division Scotland. Chlamydia Key Clinical Indicator (KCI) Year ending 31 December 2010. (Internet) 2011. Available: https://isdscotland.scot.nhs.uk/Health-Topics/Sexual-Health/Publications/2011-09-27/2011-09-27-Chlamydia-KCI-Report.pdf?42985171080 (Accessed 10 August 2014).
19. Public Health England. Sexually transmitted infections and chlamydia screening in England. Health Protection Report 2013; 8(24): 7-8.
20. Bramley G, Watkins D. Local incomes and poverty in Scotland. Developing local and small area estimates and exploring patterns of income distribution, poverty and deprivation. Report of Research for the Improvement Service and the Scottish Government (Internet) March 2013. Available: http://www.improvementservice.org.uk/assets/local-incomes-poverty-scotland.pdf (Accessed 12 April 2015).
21. Williamson LM, Scott G, Carrick-Anderson K, Kernaghan K, Hart GJ. Chlamydia trachomatis testing among 13-25 year-olds in non-genitourinary medicine settings. Journal of Family Planning and Reproductive Health Care 2007; 33(3): 177-182.
22. Adams EJ, Charlett A, Edmunds WJ, Hughes G. Chlamydia trachomatis in the United Kingdom: a systematic review and analysis of prevalence studies. Sexually Transmitted Infections 2004; 80(5): 354-362.
23. Health Protection Scotland and Information Services Division. Sexually transmitted infections and other sexual health information for Scotland (Internet). Available: http://www.documents.hps.scot.nhs.uk/bbvsti/sti (Accessed 17 July 2014).
___________________________________
Appendix I: Male and female positivity compared with test type